A study from the Centre for Heart Lung Innovation at St. Paul’s Hospital characterized the collage of ways COVID-19 damages the heart in a manner unique from other viruses.
Over 20 per cent of COVID-19 patients have some form of cardiovascular injury, and those who do have a significantly higher risk of dying from the disease. The Bruce McManus Cardiovascular Biobank (BCMB) team at the Centre for Heart Lung Innovation (HLI) investigated how the SARS-CoV-2 virus causes this damage. They examined cardiac muscle samples from 21 patients who had fatal COVID-19 and compared them to both control samples and samples from patients impacted by other viruses. Their findings, which were published in Laboratory Investigation, indicated that the way COVID-19 causes cardiac damage differs from other viruses that affect the heart, known as “cardiotropic” viruses.
Collaboration was key to the study
Dr. Bruce McManus, former director of the HLI and scholar in this area of research, has studied viral heart disease for 40 years. When the COVID-19 pandemic began, he was interested in this new human pathogen and had significant knowledge of virus-related heart disease to bring to bear. He gathered a team, including lead authors of the study Dr. Paul Hanson and Felicia Liu-Fei, to learn what they could about how this virus was damaging the heart.

The University of Nebraska Medical Center was integral to the team acquiring the COVID-19 samples. At the time, BC was not allowing autopsies of people who had COVID-19, but the University of Nebraska Medical Center has a long history of studying viral human infections like Ebola, so the protocols necessary to conduct a high-risk autopsy were already in place. The team connected with Dr. Stanley Radio, who was Dr. McManus’s clinical fellow when he was on the faculty there many years before, and Dr. Daniel Anderson, a former graduate trainee of Dr. McManus’s and current head of Cardiology at the university. They were able to obtain the samples and get them to the HLI team for study.
Mechanisms of damage
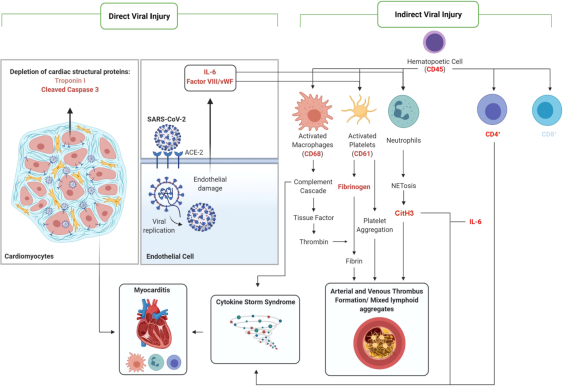
There are two broad categories by which viruses damage the heart muscle. The first is direct viral injury, in which the virus infects the heart muscle cells, or cardiac myocytes, causing the death or dysfunction of those cells. Enough cell death or damage leads to arrhythmias or heart failure. This process is the main mechanism of damage when cardiotropic viruses attack the heart.
The other category indirect viral injury, in which the damage to the heart muscle does not come from the infection of the heart cells, but from other effects caused by the virus. This study found that most of the heart damage caused by COVID-19 came from indirect viral injury.
“We were surprised, because in most viral heart disease, there is a huge amount of direct viral injury of the cardiac myocytes,” says Dr. McManus. With the SARS-CoV-2 virus, however, the majority of injury came from outsized immune activation, blood clotting, and cells dying due to a lack of oxygen. “It’s quite different from what we have seen from previously well studied heart infections by viruses.”
Role of the immune response
The immune response to the SARS-CoV-2 virus is part of what causes this damage – the level of immune activation is disproportionate to the amount of virus in the system. This increases inflammation and creates a pro-coagulative environment that causes blood clots, a major way the heart is injured.
“We need the immune response, we need the inflammatory response to take away debris and so on when viral infections occur, but the response to this virus is so immunologically robust and sort of off-kilter that it doesn’t have the same balance to it,” says Dr. McManus.
Another difference between the SARS-CoV-2 virus and other cardiotropic viruses is that it does not just target the heart.
“Viruses that damage the heart, they typically directly infect the cardiac myocytes – that’s their primary locus of injury. They’re primarily targeting the heart, not other organs, whereas COVID-19 seems to target the endothelium, which lines the vasculature and effects just about everything, including the lungs,” says Dr. Paul Hanson. Injury to the lungs is another mechanism of indirect heart injury – a patient not getting enough air due to lung damage can deplete the heart of oxygen, damaging it and other organs. The infection of the endothelium is also a large factor in the processes of blood clotting and limiting of available oxygen.
There is still more to learn
“Lots of research still needs to be done in this field, and there are endless avenues to look into to continue to further understand the virus,” says Felicia Liu-Fei.
These observations are part of a global effort to understand the behavior of the SARS-CoV-2 virus. The scale of this study by the Centre for Heart Lung Innovation was quite small, but it produced valuable insights. It will be important in the future to verify these results in the larger population.
“I thought I knew a lot about viral heart disease,” says Dr. McManus. “I think this family of SARS viruses is teaching us about a whole different major pathway for damaging a key organ like the heart.”
This study may be able to point researchers in the right direction to help to develop more diagnostic or therapeutic tools for COVID-19 associated heart damage, and could potentially help shed light on its long-term effects.
Story by Grace Jenkins





